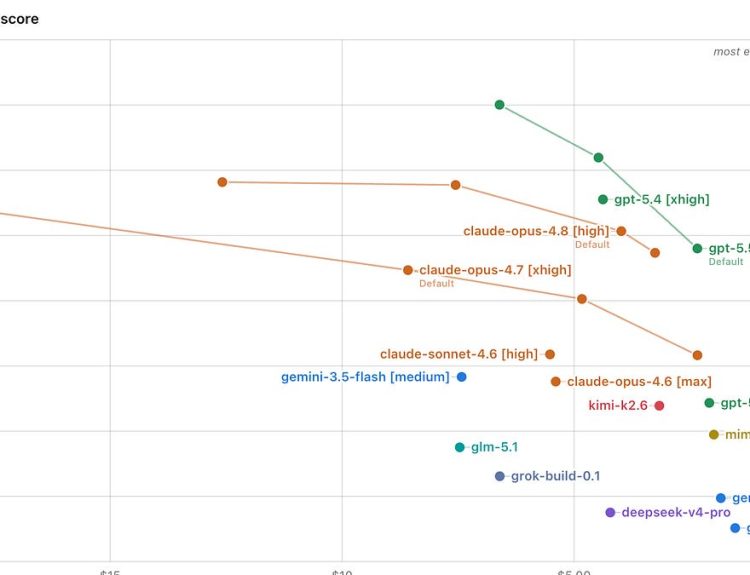
Researchers have created a machine learning system capable of predicting how intricate chemical reactions yield the appropriate molecular structure for medications.


The field of drug discovery is fundamentally about assembling atoms until a beneficial molecule is formed. This process often requires substantial investments of time and money to enhance molecular outcomes.
To address these challenges, researchers have introduced a novel machine learning system that substantially accelerates this process while reducing costs, facilitating more efficient predictions regarding how molecules assemble during chemical reactions.
“We often utilize advanced, physics-based computational chemistry tools for analyzing new reactions, but these methods are prohibitively expensive for predicting thousands of potential molecules,” explained Simone Gallarati, co-lead author of the study and a joint postdoctoral researcher at the University of Utah and the University of California. “Our goal was to develop statistical models that could accurately forecast untested reactions while keeping costs low.”
Understanding Molecular ‘Handedness’
A significant hurdle in chemistry is that molecules can appear as mirror images, a phenomenon referred to as handedness. One variant may have beneficial medical properties, while its counterpart could be ineffective or detrimental.
A core challenge in chemistry lies in the fact that molecules can exist as mirrored versions of themselves, termed handedness.
Chemists strive to design reactions that produce the intended “hand” of a molecule. This requires meticulous selection of the right combination of catalysts, ligands, and substrates.
The newly developed system functions as a sophisticated filtering mechanism, capable of evaluating tens of thousands of chemical structures and predicting how their components will amalgamate. It translates the elements of a reaction into numerical data for computer analysis, forming the foundation for machine learning predictions.
Remarkably, the model needed relatively minimal training data to demonstrate impressive performance. Even with limited information, it accurately predicted the behavior of reaction components, thus significantly cutting down the time, energy, and costs typically associated with laboratory testing.
“The majority of AI models require vast amounts of data for effective training. This poses a unique problem in chemistry, where obtaining high-quality datasets via experimentation can be both costly and time-consuming,” said Matthew Sigman, a chemist at the University of Utah and co-author of the study. “The most exciting aspect of this tool is that it enables researchers to gather smaller datasets and still build reliable models, yielding accurate predictions for known reactions and extending those predictions to untested situations.”
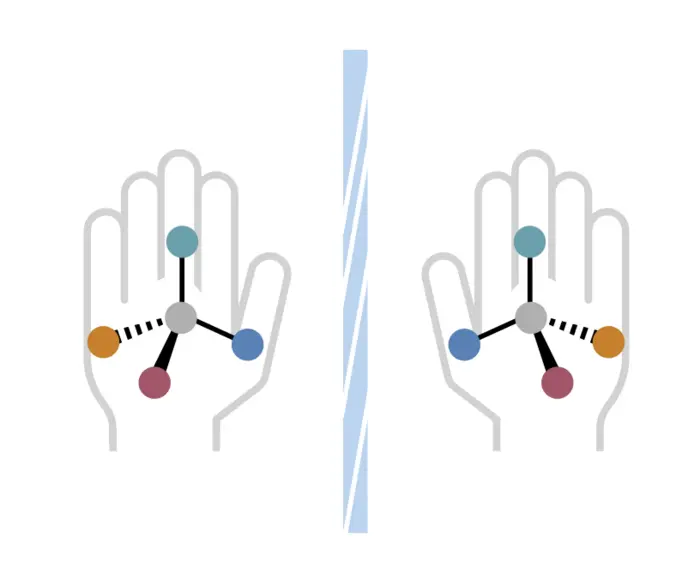

Many medicinal molecules exhibit a symmetry called handedness, featuring the same atoms arranged in the same order, but with a 3D structure that is mirrored and cannot overlap. The body reacts distinctly to “right-handed” and “left-handed” versions of these molecules. Credit: Erin Bucci/UCLA.
A Sophisticated Filter for Complex Reactions
This research concentrated on asymmetric cross-coupling reactions that are pivotal in drug development. These reactions merge two carbon-based molecular fragments using a metal catalyst to yield more intricate compounds.
These reactions are characterized as asymmetric because they are engineered to favor one version of a molecule. Without precise management, experiments could yield equal amounts of both mirrored structures. In contrast, a successful asymmetric reaction could produce 95 percent of the desired variant and only 5 percent of its unwanted mirror image.
Typically, these reactions require three main components: a metal catalyst, a ligand, and substrates. The metal catalyst facilitates the joining of carbon-based molecules, while the ligand attaches to the metal to direct which side of the molecule reacts, influencing the three-dimensional orientation of the final product. Consequently, the ligand plays a crucial role in determining the handedness of the molecule.
This tool is incredibly beneficial for chemists in the lab, significantly reducing experimental time.
To train their model, Gallarati and colleagues examined data from four academic studies on asymmetric reactions using nickel-based catalysts with various ligands. They then tasked the system with predicting outcomes for hypothetical reactions involving components it had never encountered before.
The laboratory tests for these predictions were conducted in the lab of study co-author Abigail Doyle at UCLA, with doctoral student Erin Bucci taking the lead.
“This tool is invaluable for a chemist working in a lab, it saves considerable time on experiments,” noted Bucci. “Rather than conducting 50-60 reactions, we can now manage 5-10, potentially saving weeks or even months. Each component we test requires either procurement or synthesis from scratch—this tool substantially reduces the costs associated with materials.”
Impact on the Pharmaceutical Industry
Beyond the specific nickel-based reactions explored in this study, the researchers believe their workflow could be applied widely across the field of chemistry, potentially enhancing the understanding of chemical mechanisms.
This tool allows us to glean insights about chemistry from the predictions, even when they are not accurate.
According to Sigman, the pharmaceutical industry stands to gain substantially from this approach. Drug developers often need to modify established reactions to produce the specific amounts of compounds required for clinical trials.
“This is where our tool could prove particularly useful,” Sigman emphasized. “Optimizing both the reaction procedures and their cost-effectiveness is crucial in drug development. This streamlined method could be pivotal when transitioning a molecule from Phase I to Phase II of clinical trials.”